
However, consumers should always do their due diligence and maintain realistic expectations about the benefits of using a baby monitor. Dream Sock and other innovative solutions may enhance digital monitoring and provide useful insights. But the truth is that even the fanciest and most expensive monitors cannot guarantee the safety of a sleeping baby. Since the 90s, parents have understandably turned to baby monitors out of concern about Sudden Infant Death Syndrome (SIDS) and other conditions that contribute to infant morbidity. Zenith developed the Radio Nurse in response to the 1932 kidnapping of Charles Lindberg’s infant son. There’s a dark side to baby monitoring, in that these devices were invented to help prevent unbearable tragedy. According to this notice, in a survey of 5000 parents, 94% percent reported that the Smart Sock promoted better sleep. In an initial response to the FDA letter dated October 4, 2021, Owlet states that results from 3rd-party studies have successfully demonstrated the safety of the Smart Sock. The statement points out that the FDA has not suggested that consumers using the Smart Sock should return or exchange their devices. Owlet released a statement in February 2022 explaining that the company is working with the FDA and will apply for medical device clearance before reinstating those features. The new Dream Sock does qualify as a low-risk device as it only sends notifications when the baby is awake, unable to sleep soundly, or appears to be uncomfortable. market legally.Īlthough the FDA letter didn’t mention any specific safety concerns, it cited Smart Sock’s “proactive notifications via lights and sounds if your baby’s oxygen level or heart rate leave preset zones.” Such notifications are considered diagnostic and fall outside the classification for low-risk trackers designed to promote a healthy lifestyle. 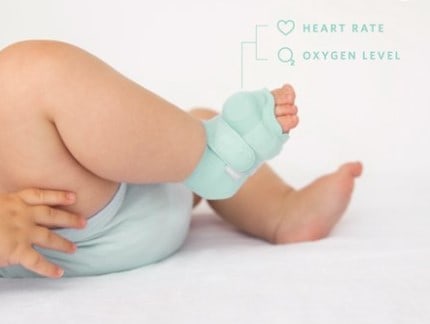
According to the FDA, the Smart Sock qualified as a medical device, yet it had not received the required FDA clearance to enter the U.S. Owlet stopped selling the Smart Sock in response to an OctoFDA warning letter informing the company that selling the Smart Sock violated federal regulations. The company introduced the Dream Sock after discontinuing its first product, the Owlet Smart Sock. has itself experienced some recent controversy. Still, for the sake of exhausted parents everywhere, we should probably hold out hope that advanced AI can bring about a non-controversial infant sleep solution.īut Owlet Baby Care, Inc. It’s easy to feel skeptical about this feature. The app also provides guidance to improve baby’s sleep: a goal that has spawned many books, techniques, and heated debates over the years. Dream Duo adds the Owlet Cam to the platform, so parents can see and hear their baby within the Owlet system. Via a companion app, parents can check the room temperature, noise levels, and humidity, and track sleep history and sleep quality. It wraps comfortably around the baby’s foot, monitoring sleep status, wakings, heart rate, and movement. Affixed with a small, circular sensor, the soft sock resembles a tiny ankle brace. The newly launched Owlet Dream Sock takes infant monitoring into the realm of wearable sleep tracking.  
These days, digital monitors with HD cameras, precision audio, and smartphone connectivity help parents relax during those precious hours when baby finally sleeps. Since then, expectant parents have poured much time and energy into determining which monitoring product can offer some peace of mind during naps and nighttime. But baby monitors didn’t become mainstream until the 1990s, when frequency hopping technology made baby monitors much more reliable. The first baby monitor, known as the Zenith Radio Nurse, was invented in 1937.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
